The Principle
The method: Analyzing isotopes
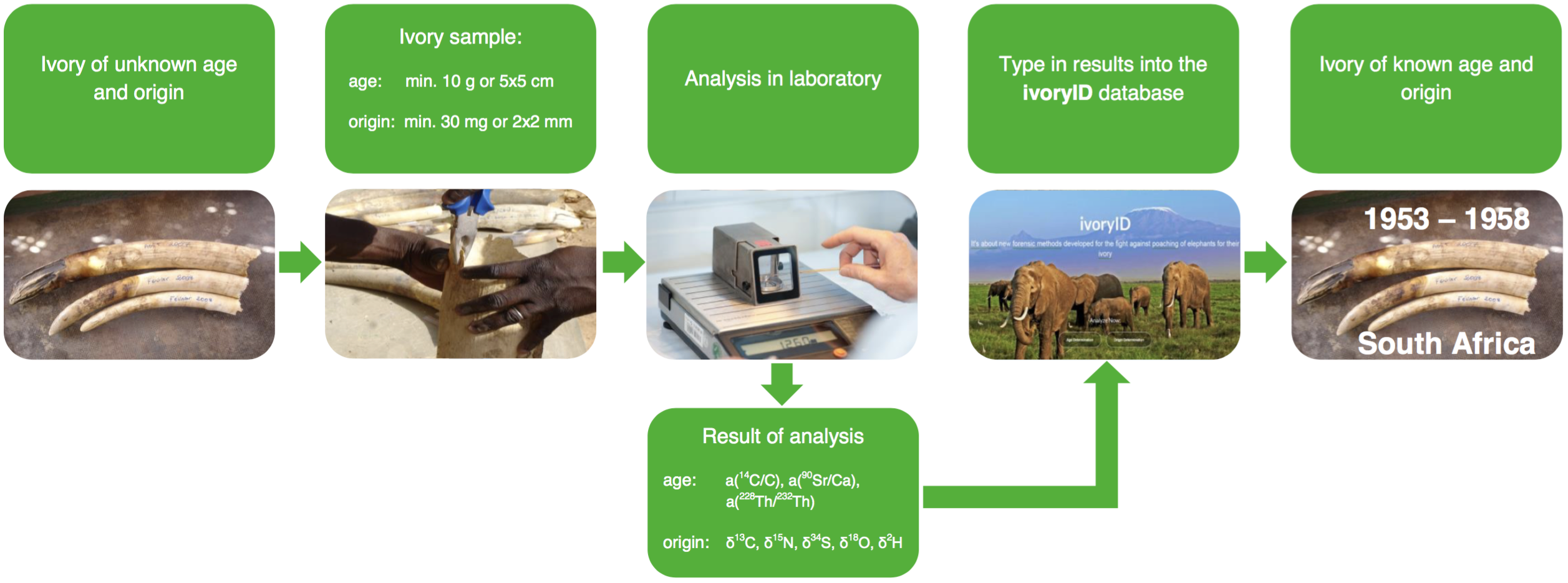
Forensic analytical methods can play an important role in conserving wild populations as well as in the investigation of wildlife crime through identification and profiling of tissue samples (Voigt et al. 2012, Moncada et al. 2012). A definitive scientific method to determine sample origin and age involves isotope analyses. Isotopes are different forms of an element that have dissimilar massing because of different neutron numbers in the nucleus. Most isotopes on earth are stable, but some are radioactive and decay over a period of time, which is characteristic of their half-life. Stable isotope analysis uses the natural abundance of isotope variation as geographic tracers, whereas radioisotopes can be used to determine the age of raw or worked elephant ivory.
Isotopes are everywhere in the environment and they are incorporated into the tissues of plants through soil and water and into animal tissues through eating, drinking and breathing (see figure below), while undergoing certain changes in isotope ratios (Hayes 1982). Throughout its life time an organism absorbs isotopes (air, water, food). They get deposited into all kind of tissues (e.g. skin, hair, bone) and usually are replaced within the cellular renewal. An exception from this rule is teeth: once formed, dental enamel does not remodel. Consequently, teeth are very useful for determining the environment and conditions of an individual's early life.
Origin and deposition of isotopes

Ivory is secreted at the margin of the inner pulp cavity of a tusk; therefore, the youngest ivory is found along this margin, becoming progressively older as the distance outwards from the margin increases. The transverse growth rate is approximately 5 mm per year and the longitudinal growth rate is approximately 5 cm per year (Uno et al. 2013). Ivory is composed of around 70% bioapatite (containing 1% carbon) and 30% collagen (containing 45% carbon). The bioapatite and collagen in ivory do not exchange elements or isotopes once formed, and therefore they record the history of diet and provide a time sequence of the isotope ratios (Codron et al. 2012).



